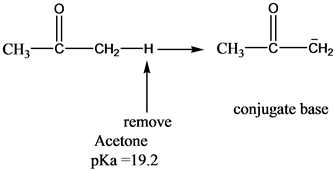Write a structural formula of the conjugate acid formed by the reaction of CH3CH2OH with HCl. | Homework.Study.com

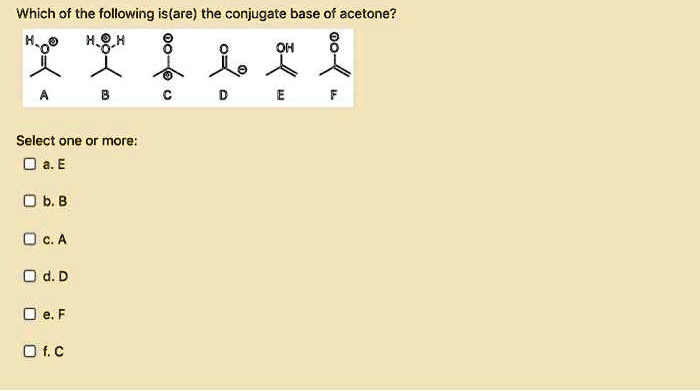
OneClass: Draw the structure of the conjugate acid of acetone (CH3COCH3) Draw the structure of the co...

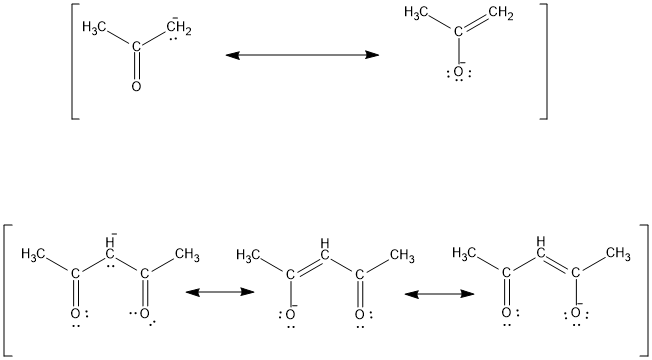
2,4-Pentanedione is a considerably stronger acid than is acetone. Write a structural formula for the conjugate base of each acid and account for the greater stability of the conjugate base from 2,4-pentanedione.

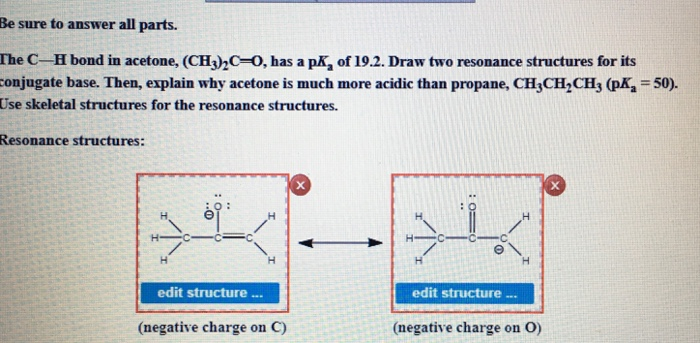
The C-H bond in acetone, (CH3_)_2C=0, has a pKa of 19.2. Draw two resonance structures for its conjugate base. | Homework.Study.com


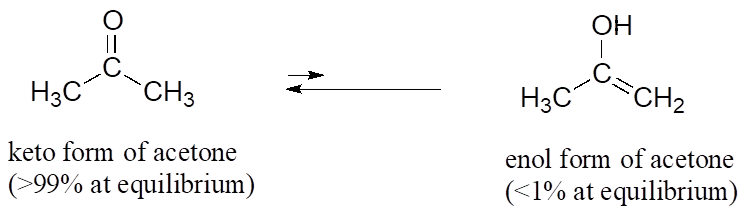
![Solved] As noted in Table 3.1, the pKa of acetone | SolutionInn Solved] As noted in Table 3.1, the pKa of acetone | SolutionInn](https://dsd5zvtm8ll6.cloudfront.net/si.question.images/image/images11/877-C-O-S(329).png)