Problem Set 2 with Solution Key - Elementary Inorganic Chemistry | HCHE 111G | Assignments Inorganic Chemistry | Docsity

An aqueous solution is prepared by diluting 3.30 mL of acetone density =0.789 g / mL to get the final volume of 75 mL. Find the mole fraction of acetone. Assume no

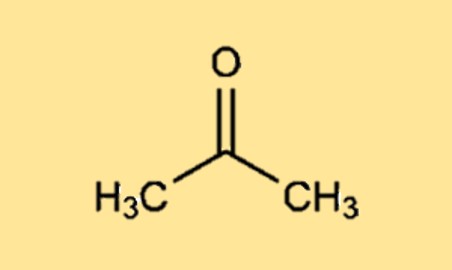
Acetone, or propanone, is an organic compound with the formula (CH3)2CO. It is the simplest and smallest ketone. It is … | Chemical structure, Chemistry, Molar mass

Calculate the volumes of acetone and ethanol that was used to mix together in order to produce 1 liter (1000 mL) of the equimolar mixture. Ethanol Mr = 46 and a density (

Tesi-2 (Cou (1) H2O (2) CH, OH (3) CHE (4) Acetone 90. The density of 1 mole of o, gas 367°C and 5 atm pressure is (1) 1 g/ml (2) 2g/ml (0) 3 g/ml (4) 4 g/ml