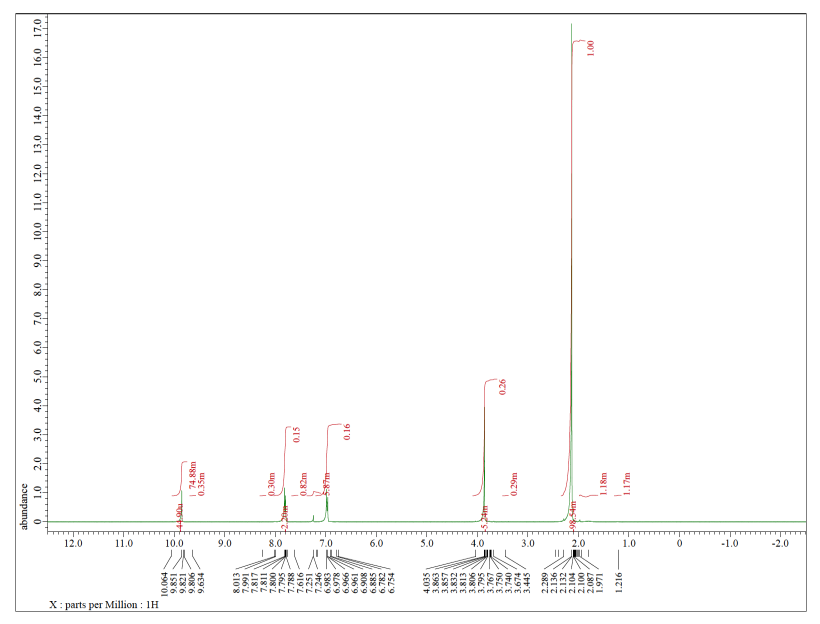
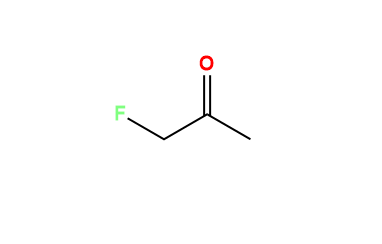
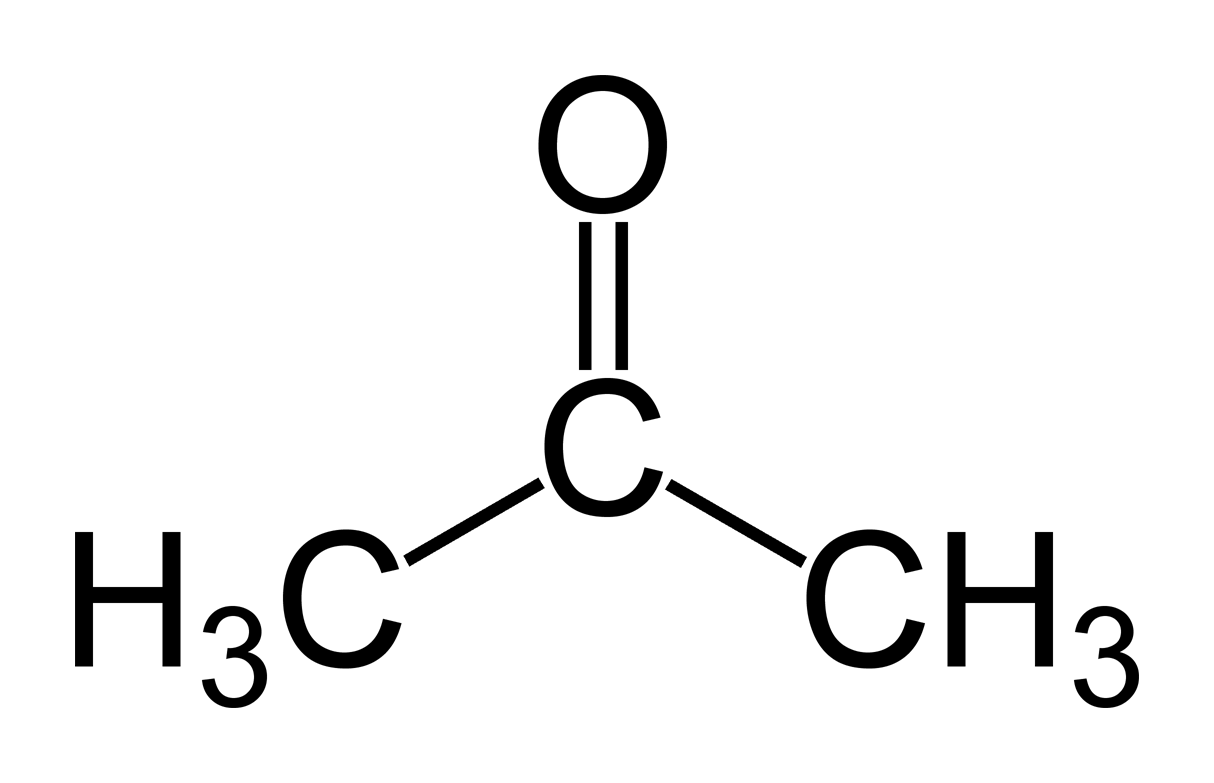
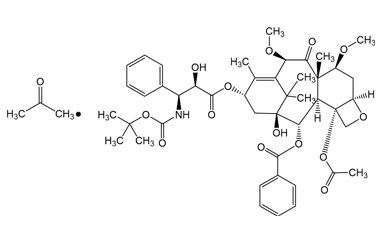
Rosuvastatin EP Impurity A (Acetone-adduct); Rosuvastatin Related compound A | EliteSynth Laboratories
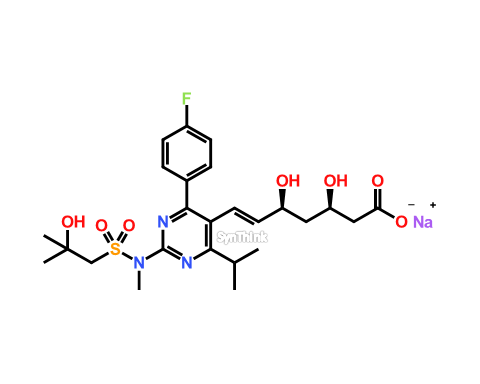
CAS: 1714147-50-8 | Rosuvastatin EP Impurity A (Acetone-adduct); Rosuvastatin Related compound A | SynThink
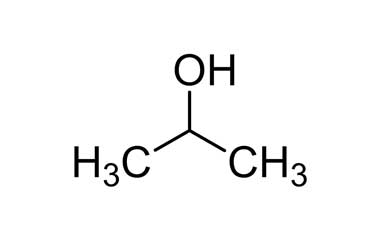
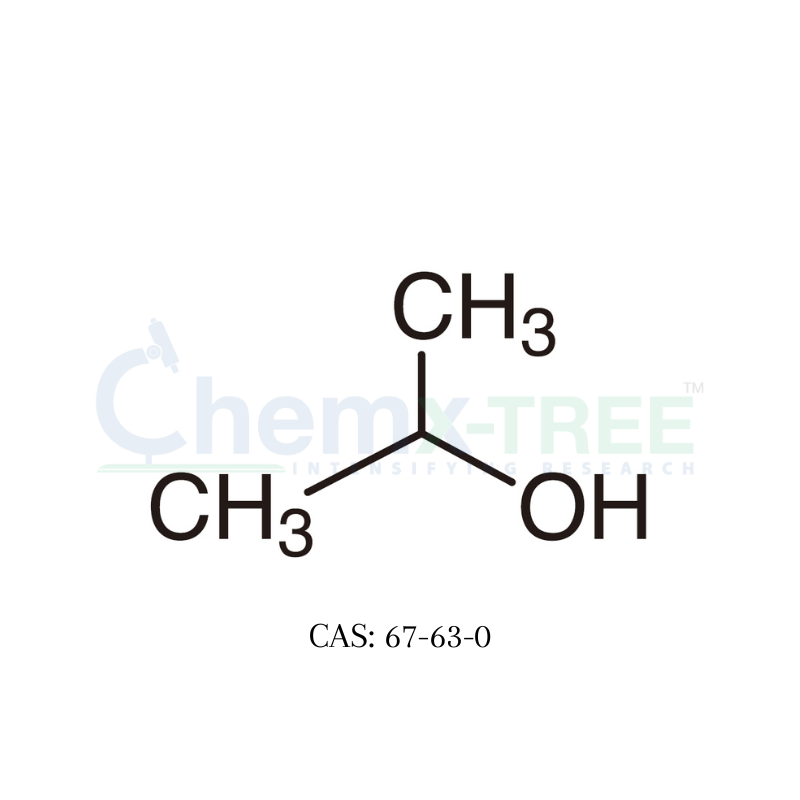
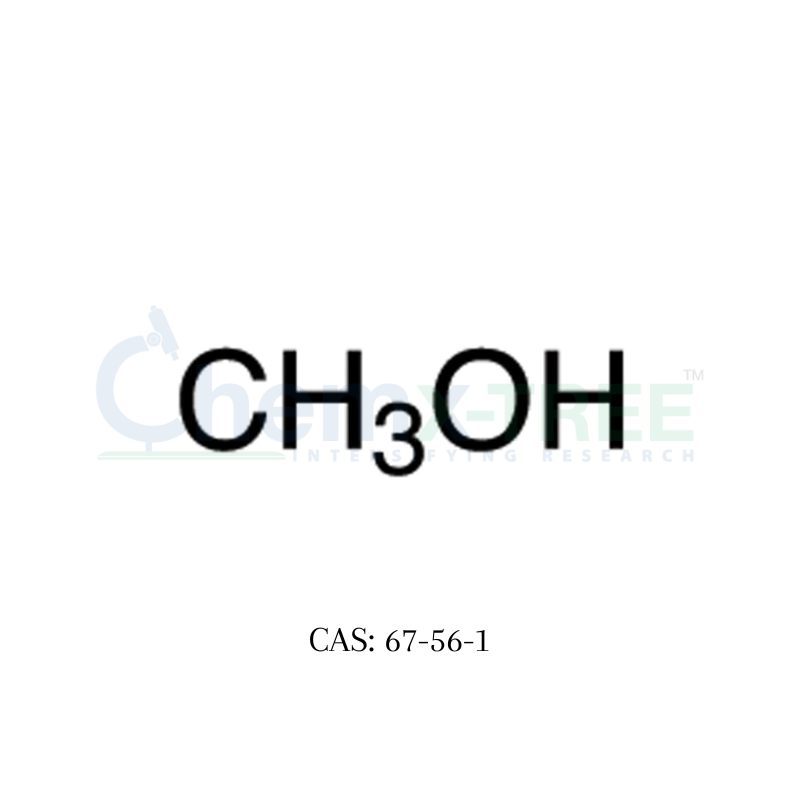
CAS No : 67-63-0 | Product Name : Acetone - Impurity B | Chemical Name : Isopropanol | Pharmaffiliates

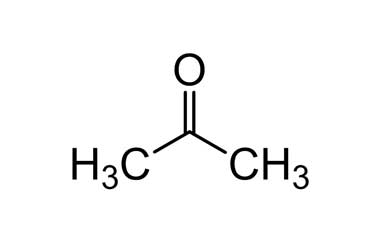
Pharmaffiliates - #Acetone is produced and disposed of in the #human_body through normal #metabolic_processes. It is miscible with water and serves as an important solvent in its own right, typically for cleaning