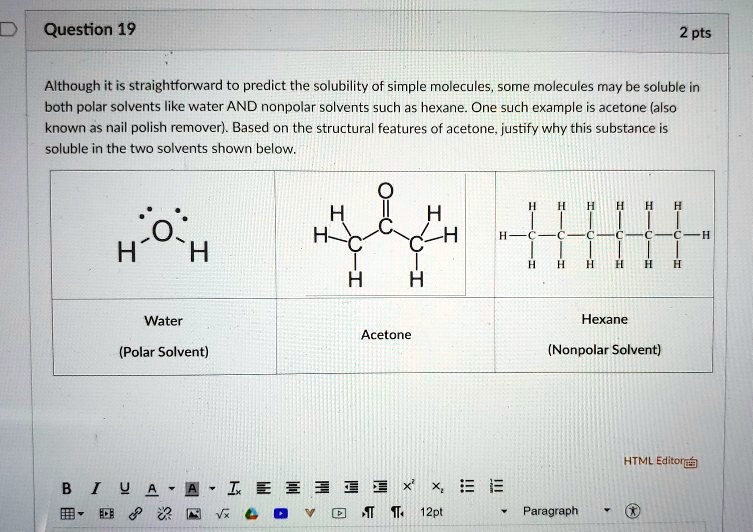
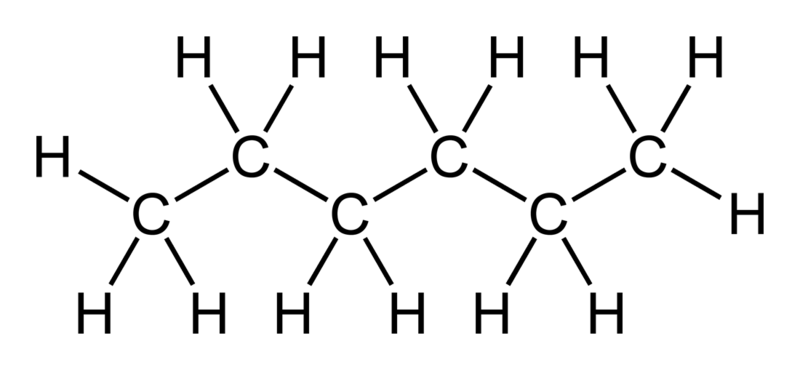
Separation of azeotropic mixture acetone + hexane by using polydimethylsiloxane membrane - ScienceDirect

Effect of solvent polarity and adsorbed water on reaction between hexyltriethoxysilane and fumed silica - ScienceDirect

Draw the structures of ethanol, acetone, toluene, hexane, and water. Classify each solvent as polar, nonpolar, or moderately polar. | Homework.Study.com

Estimation of the solubility and long-range interaction of the polymer... | Download Scientific Diagram
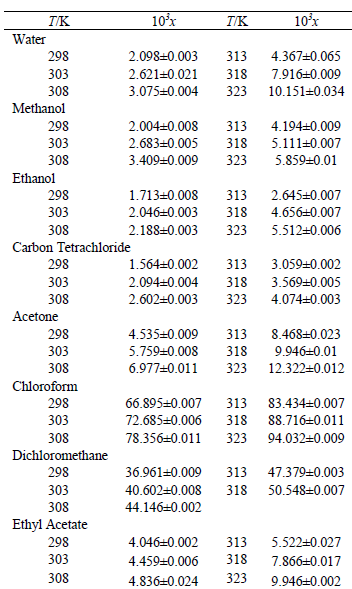
Solubility of caffeine in water, ethyl acetate, ethanol, carbon tetrachloride, methanol, chloroform, dichloromethane, and acetone between 298 and 323 K

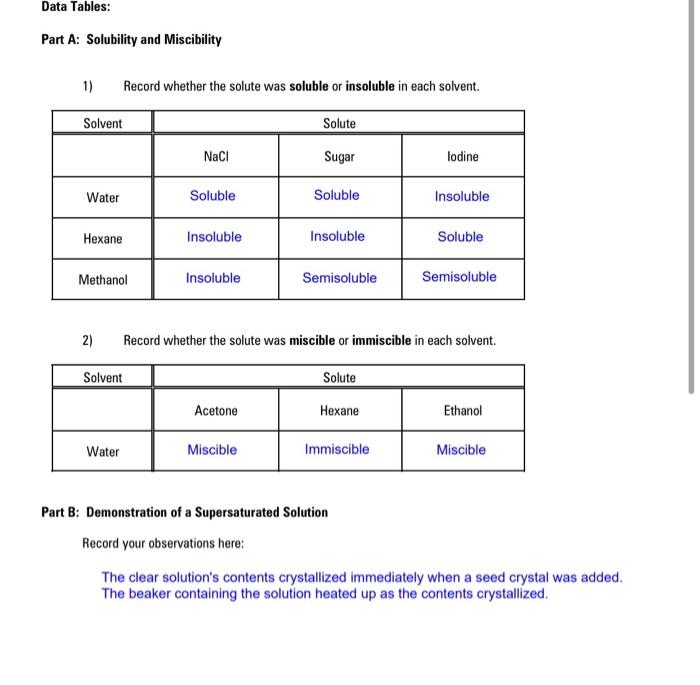
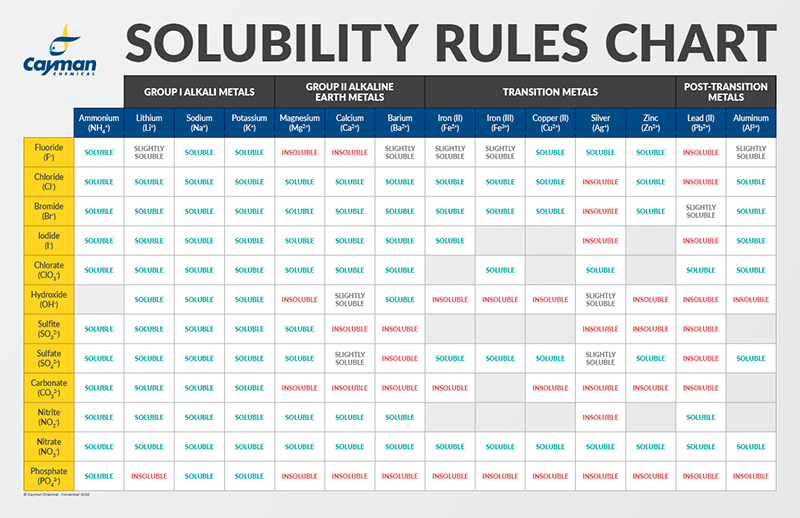
ONE Wu De Ils concentration? 23 Suggest the important of intermolecular attractive interaction the following pairs. (1) n-hexane and n-octane (11) I, and CCI, (iii) NaClo, and water (iv) methanol and acetone (

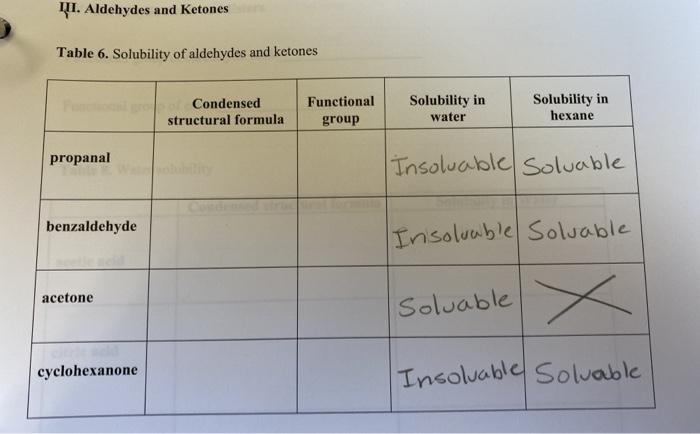
1 Why is benzoic acid insoluble in water?2 Why is acetone soluble in hexane?3 Why are methylene c - YouTube
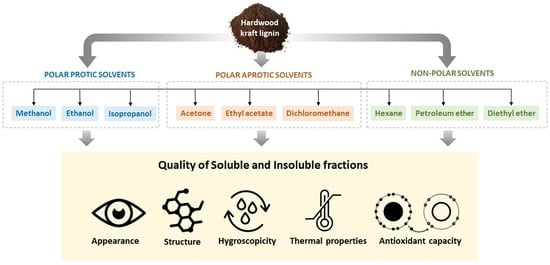
Polymers | Free Full-Text | One-Step Lignin Refining Process: The Influence of the Solvent Nature on the Properties and Quality of Fractions
![SOLVED: Acetone [(CH3)C=O] is a useful solvent because it dissolves a variety of compounds well. For example, both hexane [CH3(CH2)4CH3] and H2O are soluble in acetone. Explain why these solubility properties are SOLVED: Acetone [(CH3)C=O] is a useful solvent because it dissolves a variety of compounds well. For example, both hexane [CH3(CH2)4CH3] and H2O are soluble in acetone. Explain why these solubility properties are](https://cdn.numerade.com/ask_images/9d349811a4444b3a91bc31884ae75de9.jpg)
SOLVED: Acetone [(CH3)C=O] is a useful solvent because it dissolves a variety of compounds well. For example, both hexane [CH3(CH2)4CH3] and H2O are soluble in acetone. Explain why these solubility properties are