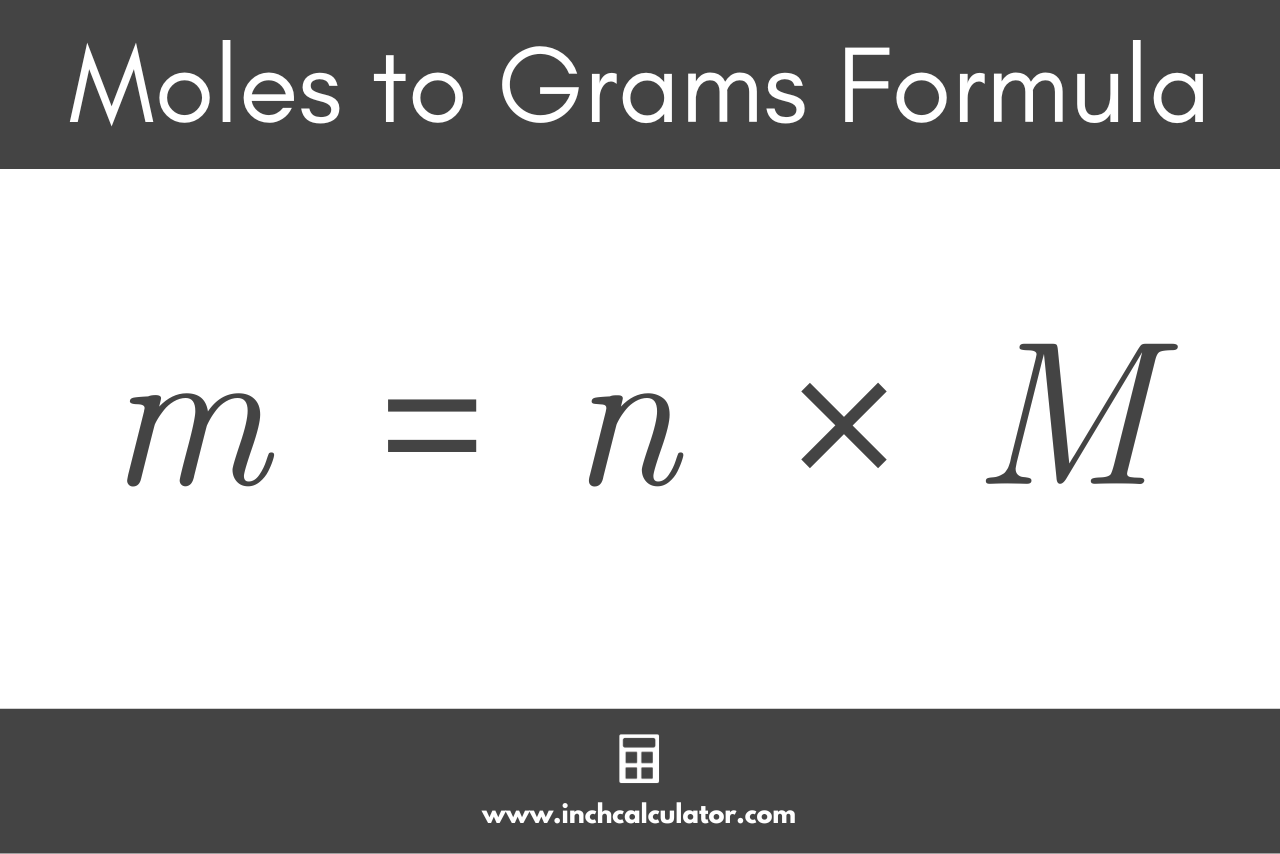Formose reaction under MW irradiation using 1.0 mol per kg formaldehyde... | Download Scientific Diagram

Conversion of a) m-X (T = 623 K, P = 0.101 MPa, W/F 0 : 25? 47 kg s mol... | Download Scientific Diagram
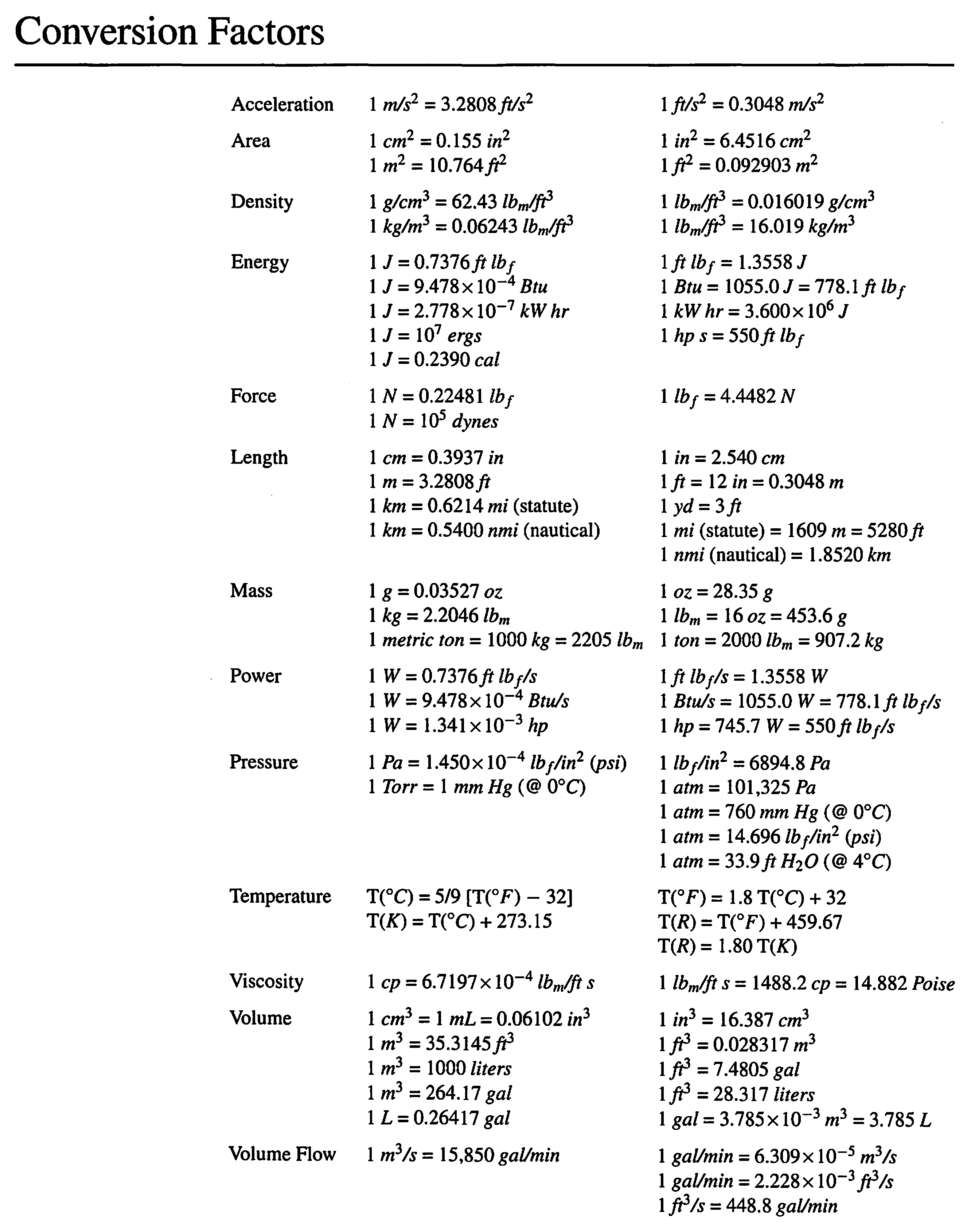
![SOLVED: Conversion Factors 1 gal = 231 in' (exact) 1 atm 760 torr (exact) cm = ] mL (exact) 1 b = 453.59237 g (exact) 2.54 cm = L in (exact) 1 SOLVED: Conversion Factors 1 gal = 231 in' (exact) 1 atm 760 torr (exact) cm = ] mL (exact) 1 b = 453.59237 g (exact) 2.54 cm = L in (exact) 1](https://cdn.numerade.com/ask_images/a1b97c8ec9ce48bcb5b41f914204e5b4.jpg)
SOLVED: Conversion Factors 1 gal = 231 in' (exact) 1 atm 760 torr (exact) cm = ] mL (exact) 1 b = 453.59237 g (exact) 2.54 cm = L in (exact) 1
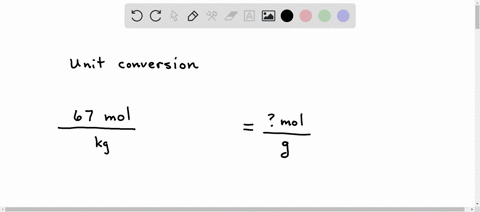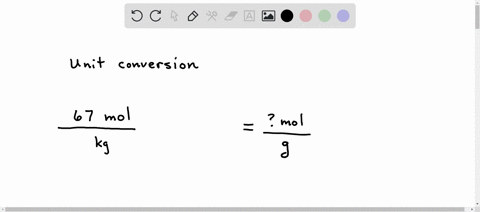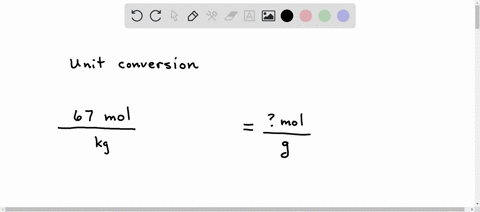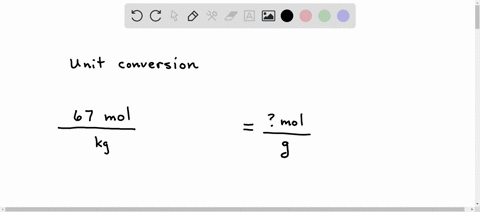
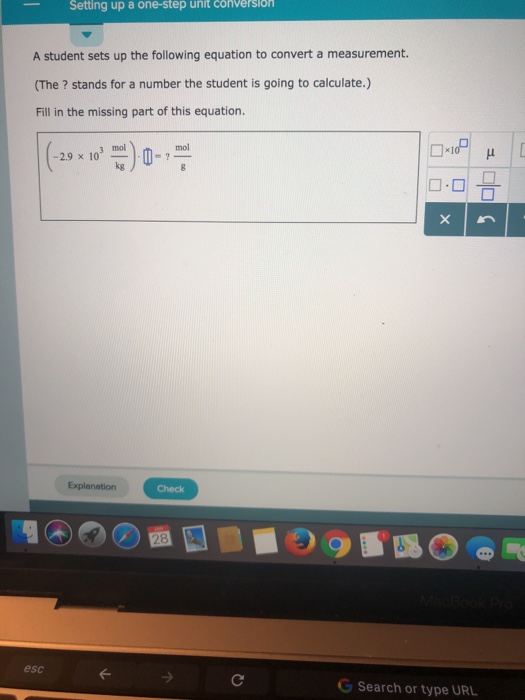
SOLVED: A student sets up the following equation to convert a measurement. (The ? stands for a number the student is going to calculate.) Fill in the missing part of this equation. (

SOLVED: Show the conversion of 1.94 mL of isopropanol to a mole value. (MW isopropanol 60.1 g/mol, density of isopropanol 786 kg/m^3). Hint: how will you deal with the conversion of mL
![SOLVED: Fundamental constants and conversion factors: NA 6.022 x1023 mol-1 k (or kB) =1381*1023 ] K-1 R=kNA =8.314] K-lmol-1 = 8.314 Pa m: mol-1 K-1 1.602 x10-19 Nae = 9.649 x104€ mol-1 SOLVED: Fundamental constants and conversion factors: NA 6.022 x1023 mol-1 k (or kB) =1381*1023 ] K-1 R=kNA =8.314] K-lmol-1 = 8.314 Pa m: mol-1 K-1 1.602 x10-19 Nae = 9.649 x104€ mol-1](https://cdn.numerade.com/ask_images/f1cfba19e7004434b3b3fc26c055851b.jpg)